SnapDx
ABSTRACT
Resource-poor regions, which often have the highest rates of treatable or preventable infectious diseases, lack access to sensitive and reliable molecular diagnostics, despite the core molecular components for diagnostics (e.g. PCR, RPA, LAMP) having existed for the past several decades. In many cases, patient samples have to be shipped to centralized laboratories to be processed. Additionally, current molecular diagnostics rely on expensive, often proprietary, reagents as well as highly technical processes, providing a barrier to LMIC in manufacturing their own diagnostics locally–a crucial requirement for building and maintaining healthcare capacity affordably in any region.
BIG QUESTION
“How do we get molecular diagnostics into the hands of community health workers who are serving the most resource-constrained areas?”
Sample to result:
under 60
minutes
Usability study:
~600
participants
Limit of detection:
1875
copies/uL
Sensitivity:
82.7
%
Specificity:
95.8
%
What is SnapDx?
SnapDx is an ultra-low cost point-of-care diagnostic device capable of running a molecular assay without any electricity, batteries, bulky lab equipment, pipetting, a formal lab space, general consumables, or extensive training to run. This self-contained device encompasses raw sample collection all the way to final colorimetric readout in under 45 minutes. SnapDx uses loop-mediated isothermal amplification (LAMP), an isothermal nucleic acid amplification assay that operates at a single temperature between 60-65°C. Developed as a rapid response to COVID-19, SnapDx has been used for detecting SARS-CoV-2 virus in patient saliva samples.
How does it work?
The simplicity of SnapDx lies in its plasticware container, which mirrors the coaxial structure of a ballpoint pen, including a refillable cartridge of reagents. A controlled volume of sample is moved across chambers via passive thermal pumps and a manual push/pull motion by hand. Throughout the assay, SnapDx remains intact and sealed within the outer tube, eliminating risk of contamination and infecting the user.
The main body (outer tube) of SnapDx contains two chambers, one of which is used for sample collection, while the other is reserved as a buffer space. After the sample is collected, extraction buffer is added directly into the same chamber, and the device is resealed. To inactivate the viruses that may be present in the sample, the device is exposed to boiling water for 3-5 minutes. During this process, the DNA contents (from host cells and virus) are released from the host cells and a small subsample is pushed through a small opening into the inner tube (plunger) of SnapDx by a thermal pressure differential. At this point, the user manually pushes the end of the plunger to expose the subsample to the LAMP reagents. The plunger is pushed until the reagents are sealed and outside of the main SnapDx body. The device is then incubated in a water bath ~63°C for 35 minutes. The colorimetric readout utilizes hydroxynaphthol blue (HNB), an azo dye that turns sky blue in positive samples and remains purple in negative.
See our Instructions for Use and Instruction Video for more details on how to operate a SnapDx device.
Partial Instructions for Use. For the full set and accompanying video - https://www.snapdx.org/
Data
For the last 2 years, we have demonstrated the limit of detection of SnapDx to be 1875 copies/uL for spiked saliva samples using inactivated SARS-CoV-2 virus. We have also tested SnapDx with clinical patient samples. Using SalivaDirect as our comparison, we determined the sensitivity and specificity of SnapDx to be 82.7% and 95.8%, respectively. The data from this study can be seen in the adjacent figure, with the blue referring to positive samples and purple for negative.
To enhance the color interpretation from the readout, there is a web application developed by Dr. Andrew D. Straw of University of Freiburg, which enhances the blue and purple hues from digital photos taken of the colorimetric results.
Colorimetric readouts from positive (first row) and negative (fourth row) SnapDx tests. Enhanced colors are shown in second and third rows for positive and negative results, respectively.
Project Status
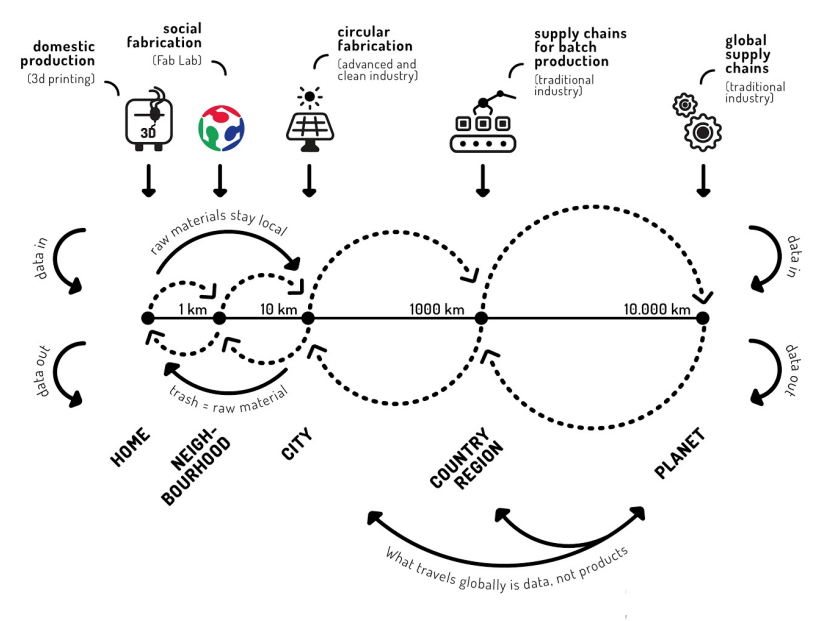
The next stage of the project will eliminate carbon-intensive cold chain shipments through local distributed biological manufacturing of reagents for the SnapDx LAMP assay. These reagents will be shelf-stable for long-term storage and be produced at a fraction of the cost of commercial, aqueous reagents while still maintaining adequate sensitivity and specificity. Further, SnapDx–a general purpose device–will be able to assess a range of samples (e.g. saliva, blood, urine) in a single platform so that low-resource areas can locally manufacture SnapDx devices that target diseases endemic to their regions, providing them autonomy and agility when responding to outbreaks.
Source: Fab City Whitepaper
Project Collaboration
The SnapDx project has engaged a number of groups within the academic and industrial sectors to both validate and manufacture the first rounds of SnapDx prototypes.
Project is funded by Stanford Medicine Catalyst (previous), Bill and Melinda Gates Foundation, and the Macromolecular Structure Knowledge Center at Sarafan ChEM-H.